 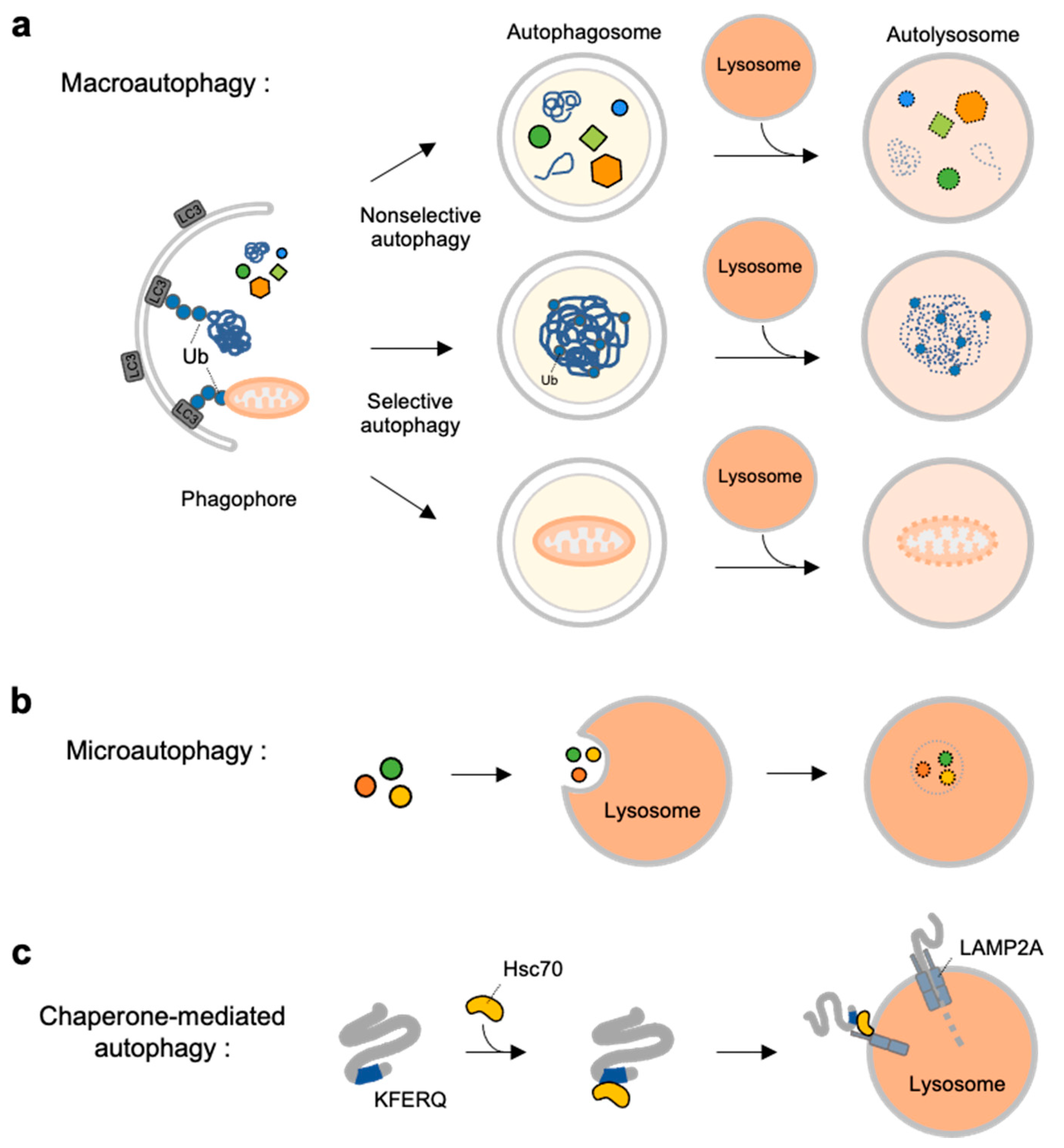
In 2012, the amyloidal nature of a class of cytosolic RIP homotypic interaction motif (RHIM)-containing proteins, receptor-interacting serine/threonine-protein kinase 1 (RIP1) and 3 (RIP3), were unveiled. 4 The term ‘functional amyloids’ was coined subsequently to distinguish these signaling amyloids from extracellularly deposited disease-related amyloids. Later, in 2009, several peptide-hormones were found to form amyloids stored in pituitary secretory granules. 1 In the early 2000s, a melanosome protein Pmel17 was found to form amyloid templates that facilitate melanin deposition during melanosome biogenesis in human melanocytes and pigment cells 2, 3 thus the Pmel17 fibril is the first human proteinaceous fibril found to fit the term ‘functional amyloids’. Surprisingly, in addition to playing devastating role in human diseases, amyloids have been found to contribute to functional entities in living organisms, such as HET-s mediating heterokaryon incompatibility and Sup35 suppressing inherited nonsense codon in fungus. They were initially defined as a distinct type of protein folding disorder that causes unspecific cellular dysfunction in a variety of diseases, including Alzheimer’s disease. The discovery that HSPA8 acts as an amyloidase dismantling functional amyloids provides a fundamental understanding of the reversibility nature of functional amyloids, a property distinguishing them from disease-related amyloids that are unbreakable in vivo.Īmyloids are a type of most well-known higher-order protein structures, all of which contain a central cross-β spine, with solvent-excluded, self-complementing steric zipper interactions. Using this amyloidase activity, HSPA8 reverses the initiator RHIM-amyloids (formed by RIP1, ZBP1, and TRIF) to prevent necroptosis initiation, and reverses RIP3-amyloid to prevent necroptosis execution, thus eliminating multi-level RHIM-amyloids to effectively prevent spontaneous necroptosis activation. Notably, HSPA8’s amyloidase activity in disassembling functional RHIM-amyloids does not require its co-chaperone system. The SBD domain of HSPA8 interacts with RHIM-containing proteins, preventing proximate RHIM monomers from stacking into functional fibrils furthermore, with the NBD domain supplying energy via ATP hydrolysis, HSPA8 breaks down pre-formed RHIM-amyloids into non-functional monomers. Different from its role in chaperone-mediated autophagy where it selects substrates containing a KFERQ-like motif, HSPA8 specifically recognizes RHIM-containing proteins through a hydrophobic hexapeptide motif N(X 1)φ(X 3). Here, we identify heat shock protein family A member 8 (HSPA8) as a new type of enzyme - which we name as ‘amyloidase’ - that directly disassembles RHIM-amyloids to inhibit necroptosis signaling in cells and mice. RHIM motif-containing amyloids, the largest functional amyloid family discovered thus far, play an important role in mediating necroptosis signal transduction in mammalian cells. However, it is unclear how these functional amyloids are effectively disassembled to terminate signal transduction. In contrast to canonical disease-related amyloids, emerging research indicates that a significant number of cellular amyloids, termed ‘functional amyloids’, contribute to signal transduction as temporal signaling hubs in humans. Ultra-stable fibrous structure is a hallmark of amyloids. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
